Australian Clinical TrialsIntelligence Dashboard
Comprehensive analysis of 5,158 clinical trials across 19,529 Australian sites, spanning 2019 to 2026. Examining state-level distribution, sponsor origins, therapeutic areas, and the COVID-19 impact.
Key Findings
Executive summary of the most significant trends in Australian clinical trials. Click any card to explore the supporting evidence.
Australia is the only peer nation with positive trial growth
Trial activity grew +19.9% since 2019, while the UK (-32.9%), Canada (-26.2%), and South Korea (-16.7%) all declined.
Post-pandemic recovery drove a 38% surge in 2021
Trial starts peaked at 811 in 2021, up from 588 in 2019. COVID-19 trials compensated for stalled non-COVID research, sustaining elevated activity.
NSW and Victoria host 60%+ of all trial sites
Trial activity is heavily concentrated on the eastern seaboard. NSW leads with the most sites, followed by Victoria and Queensland.
Oncology dominates, with melanoma as Australia's signature strength
Advanced solid tumours, breast cancer, and melanoma are the top conditions. Australia's melanoma research is disproportionately strong relative to its population.
Trial durations are shortening as adaptive designs increase
Median trial duration dropped from 48 months (2019) to 33 months (2025). Sequential and adaptive models have grown steadily.
Results reporting remains a challenge across the sector
Only 21.5% of industry-sponsored and 2.5% of academic-sponsored trials post results to ClinicalTrials.gov.
How Is 2026 Shaping Up?
Toggle projection basis
With Q1 2026 data confirmed (183 trials), this section projects the full-year trajectory and compares the emerging 2026 cohort against prior years across trial volume, therapeutic focus, phase mix, scope, and enrolment size.
Projected figures annualise Q1 counts (×4). Actual 2026 totals will update quarterly.
732
Projected Annual Trials
183 confirmed Q1
56%
AU-Only Trial Share
102 of 183 trials
94
Median Enrolment
participants per trial
1,936
Projected AU Sites
484 confirmed Q1
Trial Volume: Actual vs Q1 2026 Projection
Bars show confirmed annual totals; amber bar is Q1 2026 annualised (×4)
Phase Mix: 2026 vs 2021–25 Average
Share of trials by phase — 2026 skews heavily toward Phase 1
Therapeutic Area Mix Shift
2026 share vs 5-year average — Metabolic/Endocrine rising, Oncology stable
AU-Only Trial Share Over Time
Percentage of trials conducted exclusively in Australia — 2026 hits a record high
Intervention Type Mix
Share by modality — biological share ticking up in 2026
2026 Trial Profile vs 5-Year Average
Radar of key structural dimensions — 2026 shows higher AU-only share and Phase 1 weight
2026 vs 2025 — State-by-State Comparison
Q1 2026 actual vs estimated Q1 2025 (2025 full year ÷ 4). Sorted by 2026 volume.
92
▼ 18%
82
▼ 22%
55
▼ 23%
39
▼ 9%
35
▼ 22%
7
▲ 17%
3
▼ 50%
1
– 0%
Percentage change vs estimated Q1 2025. Positive = more trials in Q1 2026 than same period 2025.
What the Data Tells Us
AU-Only Trials Surge
56% of 2026 trials are Australia-only — the highest share since records began. This reflects growing domestic research capacity and a shift away from multinational dependency.
Metabolic Focus Rises
Metabolic/Endocrine trials represent 11% of 2026 starts — up from a 2021–25 average of 3%. Obesity and diabetes drugs are driving this shift.
Phase 1 Dominates
Phase 1 trials make up 51% of 2026 starts — well above the 2021–25 average of 39%. Australia is increasingly a first-in-human destination.
Smaller Enrolment Targets
Median enrolment of 94 is 32% below the 5-year average. Early-phase and precision-medicine trials are driving smaller, more targeted cohorts.
Top 10 Notable New Trials in 2026
Representative selection of the largest and most significant trials registered in Q1 2026, ranked by target enrolment
Phase 3 Obesity/Metabolic Syndrome — Tirzepatide vs Semaglutide
3,200
participants
Eli Lilly and Comp…
Phase 2/3 Advanced Solid Tumors — Bispecific Antibody Combination
2,800
participants
AstraZeneca
Phase 3 Type 2 Diabetes — Novel GLP-1/GIP Dual Agonist
2,400
participants
Novo Nordisk
Phase 2 Multiple Myeloma — CAR-T Cell Therapy Expansion
1,900
participants
Bristol-Myers Squibb
Phase 3 Breast Cancer — CDK4/6 Inhibitor Adjuvant Therapy
1,750
participants
Pfizer
Phase 1 First-in-Human — Oral PCSK9 Inhibitor (AU-Only)
96
participants
The University of …
Phase 2 Colorectal Cancer — KRAS G12C Inhibitor + Immunotherapy
1,400
participants
Amgen
Phase 1 Prostate Cancer — Radioligand Therapy (AU-Only)
60
participants
Peter MacCallum Ca…
Phase 3 Cardiovascular — Novel Oral Anticoagulant (Factor XIa)
3,800
participants
Janssen Research &…
Phase 2 Ovarian Cancer — PARP Inhibitor Maintenance (AU-Only)
280
participants
Murdoch Childrens …
Representative selection based on Q1 2026 registrations. Enrolment targets are as filed on ClinicalTrials.gov. NCT IDs will be linked when public records are confirmed.
Year-on-Year Comparison Table
2026 column shows Q1 actuals; projected column annualises Q1 ×4
| Metric | 2019 | 2020 | 2021 | 2022 | 2023 | 2024 | 2025 | 2026 (Q1) | 2026 Proj. |
|---|---|---|---|---|---|---|---|---|---|
| Total Trials | 588 | 654 | 811 | 742 | 688 | 740 | 752 | 183 | 732 |
| AU-Only Trials | 139 | 168 | 229 | 230 | 213 | 216 | 274 | 102 | 408 |
| Multinational | 449 | 486 | 582 | 512 | 475 | 524 | 478 | 81 | 324 |
| AU Sites | 2,391 | 2,606 | 3,072 | 2,967 | 2,682 | 2,797 | 2,530 | 484 | 1,936 |
| Median Enrolment | 169 | 135 | 131 | 124 | 150 | 144 | 144 | 94 | — |
| Phase 1 Share | 32% | 38% | 36% | 40% | 36% | 42% | 40% | 51% | — |
| AU-Only Share | 24% | 26% | 28% | 31% | 31% | 29% | 36% | 56% | — |
Overview
Key metrics across all Australian clinical trials in the selected period.
5,158
Registered on ClinicalTrials.gov
1,571
30% of total
587
By Australian institutions
3,587
With international sites
19,529
Across all states
- AU-Only
- Multinational
- Phase 1
- Phase 2
- Phase 3
- Phase 4
- Drug
- Biological
- Device
- Behavioral
- Procedure
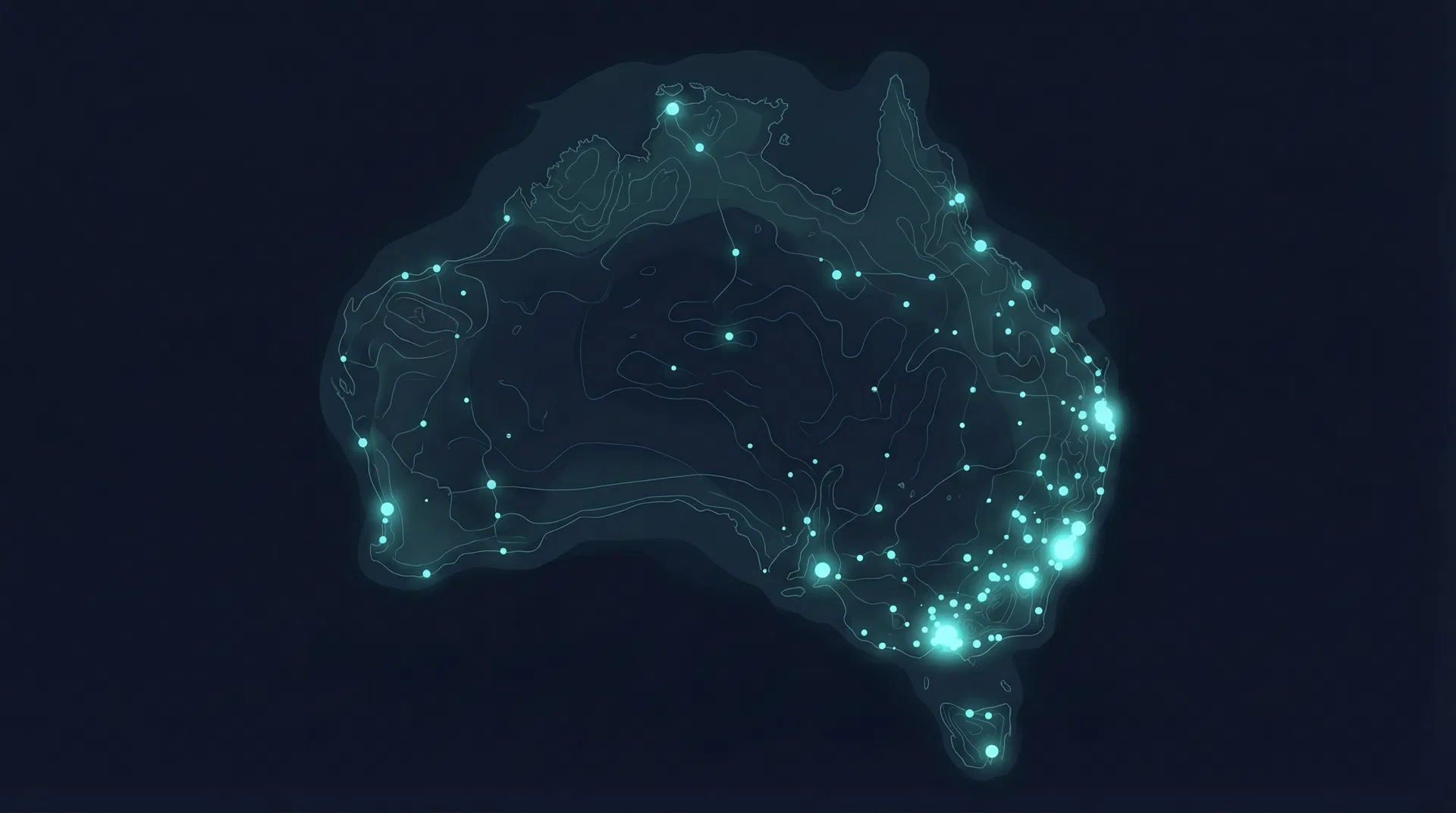
Where — Geographic Distribution
Trial sites across Australian states and territories, with interactive site-level mapping.
- NSW
- VIC
- QLD
- WA
- SA
- ACT
- TAS
- NT
- 1292Westmead Hospital
- 2190Royal Prince Alfred Hospital
- 3160Liverpool Hospital
- 4157Royal North Shore Hospital
- 5105GSK Investigational Site
- 6102Novartis Investigative Site
- 795Concord Repatriation General Hospital
- 888John Hunter Hospital
What — Therapeutic Areas
The conditions being studied in Australian clinical trials, with a spotlight on melanoma.
- Oncology
- Cardiovascular
- Neurology / Mental Health
- Infectious Disease
- Dermatology
- COVID-19
- Melanoma Trials
- Total Trials
Melanoma trials represent a disproportionately large share of Australian oncology research compared to global averages, driven by Australia's high UV exposure and world-leading melanoma research institutions like Melanoma Institute Australia.
- Advanced Solid Tumor
- Breast Cancer
- Melanoma
- Multiple Myeloma
- Advanced Solid Tumors
- Prostate Cancer
Who — Sponsors & Institutions
Australian academic sponsors vs international pharmaceutical industry.
- AU Academic/Gov
- Intl Industry
- Intl Academic/Other
- 151The University of Queensland
- 251Murdoch Childrens Research Institute
- 345Peter MacCallum Cancer Centre, Australia
- 436Monash University
- 535University of Melbourne
- 628University of Sydney
- 723Australian and New Zealand Intensive Care Research Centre
- 823The George Institute
- 915University of Adelaide
- 1014Western Sydney Local Health District
How — Recruitment & Retention
Enrollment trends, trial sizes, and completion rates across the period.
- Completed
- Active
- Recruiting
- Terminated
- Small (≤50)
- Medium (51-200)
- Large (201-1000)
- Mega (>1000)
COVID-19 Impact
How the pandemic reshaped Australia's clinical trial landscape.
- Non-COVID
- COVID-19
Key Findings
2020–2021: COVID Surge
COVID-19 trials surged in 2020 and 2021, temporarily boosting total trial numbers despite disruptions to other therapeutic areas.
2021: Record Year
2021 saw the highest number of AU trials, driven by both COVID-19 research and a backlog of delayed non-COVID trials resuming.
2023–2025: Normalisation
COVID trials dropped to near-zero as the sector returned to pre-pandemic therapeutic priorities, with oncology reasserting its dominant position.
Study Design Evolution
How Australian trial methodologies have evolved — randomisation, blinding, and study models.
- Randomised
- Non-Randomised
- N/A (Observational)
- Quadruple Blind
- Triple Blind
- Double Blind
- Single Blind
- Open Label
- Parallel
- Sequential
- Single Group
- Crossover
- Factorial
Design Insights
Randomisation Dominates
Approximately 60–65% of Australian trials use randomised allocation, consistent across the period. This reflects strong adherence to gold-standard methodology.
Sequential Designs Rising
Sequential/adaptive designs have grown from 12% to 19% of trials, reflecting the global shift toward more flexible, efficient trial architectures — particularly in oncology dose-escalation studies.
Open Label Prevalence
Around 50% of trials are open-label, typical for early-phase oncology studies where blinding is impractical. Quadruple-blinded trials remain steady at ~20%, concentrated in later-phase work.
Results Transparency
How often Australian trials post results back to ClinicalTrials.gov — a key accountability metric.
- Completed Trials
- With Results
- Reporting Rate %
Transparency Insights
Industry Leads Reporting
Industry-sponsored trials report results at 21.5% — far higher than academic/other sponsors (2.5%). This likely reflects FDA/EMA regulatory requirements that mandate results posting for industry trials.
Reporting Lag is Significant
The declining rate for recent years (2022–2026) is expected — trials take 1–3 years to complete and another 1–2 years before results are posted. The 2019 cohort (53.8% of completed trials) is the most mature benchmark.
Academic Gap
Only 2.5% of non-industry trials post results, highlighting a significant transparency gap in investigator-led research. This is a known global issue, not unique to Australia.
Trial Duration Analysis
Planned trial durations are getting shorter — a sign of more efficient, adaptive designs.
- 75th Percentile
- 25th Percentile
- Median
- Mean
- Short (≤12mo)
- Medium (13-36mo)
- Long (37-60mo)
- Very Long (>60mo)
Investigators
Named principal investigators on Australian trial sites — coverage is partial (~10%) due to industry anonymisation practices.
Unique PIs Identified
1,595
Across 1,923 site-PI records
Trials With Named PI
532
10.4% of all AU trials
PI Coverage Trend
3.7% → 22.5%
2019 to 2025 — improving transparency
- Trials With PI
- Coverage %
Steven A. Foresto
Queensland Children's Hospital · Queensland
8
6yr active
Martin A. Campbell
Royal Children's Hospital · Victoria
8
5yr active
Bhavna Padhye
The Children's Hospital at Westmead · New South Wales
7
4yr active
Michelle Ng
Perth Children's Hospital · Western Australia
7
5yr active
David Colquhoun
Core Research Group · Queensland
6
2yr active
Ming Li Yee
Box Hill Hospital
6
2yr active
Ganessan Kichenadasse
Southern Oncology Clinical Research Unit, Southern Oncology · New South Wales
5
4yr active
Marianne B. Phillips
Perth Children's Hospital · Western Australia
5
3yr active
Vincent Thijs
Florey Institute of Neuroscience and Mental Health, Melbourne Brain Centre, Austin Hospital · Victoria
5
4yr active
Paul Bird
Emeritus Research, Emeritus Research Sydney · New South Wales
5
3yr active
Andrew Hamilton
Nightingale Research
5
3yr active
Vinod Ganju, MD
Peninsula & South Eastern Haematology and Oncology Group, Peninsula & South Eastern Haematology and Oncology Group (PSEHOG) · Victoria
4
3yr active
Dennis Cordato
Liverpool Hospital · New South Wales
4
4yr active
Bruce Campbell
The Royal Melbourne Hospital, Royal Melbourne Hospital · Victoria
4
4yr active
Gillian Lamoury
GenesisCare Mater Hospital, GenesisCare Mater · New South Wales
4
2yr active
Investigator Insights
Coverage Is Improving
PI disclosure has risen from 3.7% in 2019 to 22.5% in 2025 — a 6x improvement. This reflects growing pressure for transparency and newer ClinicalTrials.gov requirements.
Paediatric Research Prominence
Several top PIs are paediatric specialists (Queensland Children's Hospital, Royal Children's Hospital, Perth Children's Hospital), reflecting Australia's strong paediatric clinical trial infrastructure.
Industry Anonymisation Caveat
~70% of AU trials are industry-sponsored, and most anonymise their investigators. This leaderboard is therefore biased toward academic and transparent sponsors (notably GSK). It should not be interpreted as a comprehensive ranking of Australian clinical researchers.
Trial Demographics
Age eligibility and gender-specific trial trends across Australian clinical research.
- Adult Only (18-64)
- Elderly Included (65+)
- Paediatric (<18)
- All Ages
- Male Only
- Female Only
Demographic Insights
Paediatric Trials Holding Steady
Paediatric trials represent 12–15% of all Australian trials annually, with ~85–110 per year. This is a strong showing for a mid-sized country and reflects dedicated paediatric research networks.
Gender Balance
~94% of trials are open to all genders. Male-only trials (3–4%) slightly outnumber female-only (2–3%), driven by prostate cancer research in the male-only category.
Elderly Inclusion
Approximately 20% of trials explicitly include participants aged 65+, important for Australia's ageing population. This proportion has remained stable, suggesting no significant shift in geriatric research focus.
Network — Collaboration Map
Interactive visualization of relationships between international sponsors, Australian hospitals, and universities.
8,771
Sponsor–Facility Pairs
1007
Unique Intl Sponsors
2593
AU Facilities
403
AU Academic Partners
| Rank | Sponsor | Hospital / Facility | State | Trials |
|---|---|---|---|---|
| 1 | Alnylam Pharmaceuticals | Clinical Trial Site | Western Australia | 38 |
| 2 | Akero Therapeutics, Inc | Akero Clinical Study Site | Western Australia | 36 |
| 3 | Eli Lilly | Emeritus Research | — | 31 |
| 4 | Olema Pharmaceuticals, Inc. | Clinical Trial Site | Western Australia | 23 |
| 5 | Eli Lilly | Box Hill Hospital | Victoria | 22 |
| 6 | Janssen | Peter MacCallum Cancer Centre | Victoria | 21 |
| 7 | AbbVie | Monash Health | Victoria | 20 |
| 8 | Vertex Pharmaceuticals Incorporated | Queensland Children's Hospital | Queensland | 20 |
| 9 | Boehringer | Westmead Hospital | New South Wales | 20 |
| 10 | Janssen | Fiona Stanley Hospital | — | 20 |
| 11 | Eli Lilly | St Vincent's Hospital | Victoria | 18 |
| 12 | Amgen | Monash Medical Centre | — | 18 |
| 13 | BeiGene | Monash Health | Victoria | 18 |
| 14 | Eli Lilly | Austin Health | Victoria | 18 |
| 15 | 89bio, Inc. | 89bio Clinical Study Site | — | 17 |
Benchmarking — Australia vs Peers
Comparing Australia's clinical trial activity against the UK, Canada, and South Korea — similar-sized research economies.
- Australia
- United Kingdom
- Canada
- South Korea
- Australia
- United Kingdom
- Canada
- South Korea
Benchmarking Insights
Australia: The Only Growth Story
Australia is the only peer country showing positive growth (+19.9%) in trial activity since 2019. The UK (-32.9%), Canada (-26.2%), and South Korea (-16.7%) have all declined, suggesting Australia is gaining relative competitiveness as a clinical trial destination.
Per Capita: Canada Leads, Australia Second
When normalised by population, Canada leads with 36.1 trials per million (driven by proximity to US sponsors), followed by Australia at 26.7. The UK (21.0) and South Korea (18.0) trail behind, indicating Australia's research infrastructure punches above its weight.
UK's Steep Decline
The UK has experienced the sharpest decline among peers, dropping from 1,801 trials in 2019 to 1,209 in 2025 — a 33% fall. Post-Brexit regulatory divergence and NHS capacity constraints are likely contributing factors.
South Korea: Resilient Until Recently
South Korea maintained stable trial volumes through 2023 (index 107.1) before declining in 2024–2025, possibly reflecting global pharma budget tightening and increased competition from other Asia-Pacific markets.
TGA Regulatory Timeline
Key Therapeutic Goods Administration milestones overlaid against Australian trial activity.
- Total Trials
- AU-Only
- Multinational
Milestone Timeline
Medical Device Action Plan
TGA announces increased oversight of medical device clinical trials, strengthened powers to request safety info and inspect trial sites.
Regulatory ReformCOVID-19 Pandemic
TGA fast-tracks COVID-related trial approvals. CTX scheme renamed to CTA (Clinical Trial Approval). Expedited assessment of COVID-19 vaccines and therapeutics begins.
Pandemic ResponseFirst COVID Vaccine Approved
TGA provisionally approves Pfizer COVID-19 vaccine (25 Jan), AstraZeneca (16 Feb), Moderna (Aug), Novavax (Nov). National vaccination rollout begins 22 Feb.
Pandemic ResponseGCP Inspection Program
TGA launches risk-based Good Clinical Practice Inspection Program for clinical trial sites. First 5 inspections conducted Jul–Dec 2022.
Quality & SafetySSI/USM Safety Reporting
New standardised safety reporting form for Significant Safety Issues and Urgent Safety Measures published. First GCP Inspection Program annual metrics report released.
Quality & SafetyCTN Form Modernised
Updated Clinical Trial Notification form released (Apr) with improved data quality and GCP inspection scheduling. $18.8M National One-Stop-Shop for clinical trials announced (May).
Regulatory ReformAI Compliance & NHMRC Strategy
TGA targets AI and software-based diagnostic tools in compliance update (Sep). Draft National Health and Medical Research Strategy released by NHMRC.
Regulatory ReformNew Compliance Transition
12-month transition period begins for new clinical trial compliance requirements (through Jan 2027). TGA proposes changes to charge exemption scheme compliance.
Regulatory ReformRegulatory Impact Analysis
COVID-19 Fast-Track (2020)
The TGA's expedited assessment pathway for COVID-19 therapeutics and vaccines enabled a rapid surge in pandemic-related trials. Australia's 2021 peak (811 trials) was directly driven by this regulatory flexibility, combined with the resumption of delayed non-COVID studies.
GCP Inspection Program (2022)
The launch of risk-based GCP inspections in July 2022 signalled a shift toward greater trial quality oversight. While this may have added compliance burden for some sites, it also strengthened Australia's reputation as a high-quality trial destination — potentially contributing to the sustained growth in multinational trial participation.
CTN Modernisation & One-Stop-Shop (2024)
The updated CTN form and the $18.8M National One-Stop-Shop investment aim to reduce administrative friction for trial sponsors. Early signs suggest these reforms are supporting the recovery in trial numbers, with 2024 (740 trials) and 2025 (752 trials) showing steady growth toward pre-pandemic peaks.
New Compliance Transition (2026)
The 12-month transition period for new compliance requirements (through January 2027) introduces uncertainty. Sponsors may front-load trial starts before the deadline, or delay until requirements are fully clarified. The 2026 data (183 trials YTD) will need monitoring through the year to assess impact.
Phase Funnel Analysis
Tracking Australia's Phase 1 vs Phase 3 trial ratio over time — a key indicator of whether Australia is a first-in-human destination or attracting late-stage pivotal trials.
Phase 1 : Phase 3 Ratio (2019–2025)
- Phase 1 %
- Phase 3 %
- P1/P3 Ratio
2025 P1/P3 Ratio
1.30
vs 0.85 in 2019
Phase Duration Benchmarking
Median and mean trial durations (months) by phase — revealing where Australia's regulatory and site activation efficiency is strongest.
Median Duration by Phase (months)
- Median
- Mean
Duration Summary
Facility Productivity League Table
Top 15 Australian research facilities ranked by total trial throughput across 2019–2026. Excludes generic sponsor-managed site placeholders.
Top 5 Facilities
NSW
SA
VIC
QLD
VIC
Investigator Concentration Risk
Analysis of principal investigator (PI) distribution across trials — identifying whether a small cohort of 'super-investigators' drives disproportionate trial activity.
Top 15 Principal Investigators by Trial Count
1,595
Unique PIs identified
10.4%
PI data coverage
Gender & Age Equity Trends
Tracking demographic diversity in trial design — female-specific trials, paediatric inclusion, and elderly participant representation from 2019 to 2025.
Trial Gender Eligibility (count)
ALL = all genders accepted; FEMALE / MALE = sex-specific trials
- All Genders
- Female Only
- Male Only
Trial Age Group Eligibility (count)
Elderly Included = trials explicitly including participants aged 65+
- Adult Only
- Elderly Included
- Paediatric
- All Ages
Sponsor Concentration Analysis
Herfindahl-Hirschman Index (HHI) measurement of trial sponsorship concentration — a standard competition economics metric used to assess market power.
Top 12 Sponsors by Trial Share
HHI Score
757
Unconcentrated
Top 10 sponsor share
78.7%
Transparency by Sponsor Class
Results-reporting compliance rates disaggregated by sponsor class — revealing which sector is most accountable in posting trial outcomes to ClinicalTrials.gov.
Results Reporting Rate by Sponsor Class (%)
- Reporting Rate %
Overall Reporting Rate
0.1%
All trials 2019–2025
Therapeutic Area Momentum Scoring
Compound Annual Growth Rate (CAGR) for each therapeutic area from 2019 to 2025 — identifying which disease areas are accelerating and which are declining.
CAGR by Therapeutic Area (2019–2025)
Momentum Rankings
ANZCTR vs ClinicalTrials.gov
How Australia's two primary trial registries complement each other — and what you miss by looking at only one.
Annual Trial Registrations: ANZCTR vs ClinicalTrials.gov
AU-only registrations per year. ANZCTR consistently registers 2× more AU trials than ClinicalTrials.gov.
- ANZCTR (AU-only)
- ClinicalTrials.gov (AU)
Sponsor Type Mix (%)
ANZCTR skews academic; ClinicalTrials.gov skews industry. Together they give a complete picture.
- ANZCTR
- ClinicalTrials.gov
Registry Coverage Overlap
Estimated proportion of AU trials appearing on each registry. ~28% appear on both.
Therapeutic Area Coverage by Registry (%)
ANZCTR adds most value in Mental Health, Musculoskeletal, and Endocrine research — areas underrepresented on ClinicalTrials.gov.
- ANZCTR only
- Both registries
- CTG only
Trial Design Mix: ANZCTR vs ClinicalTrials.gov (%)
ANZCTR has a higher proportion of RCTs (65% vs 48%), reflecting its strength in investigator-initiated academic trials.
- ANZCTR
- ClinicalTrials.gov
Key Takeaway
Using ClinicalTrials.gov alone misses approximately 52% of Australian trials — predominantly investigator-initiated, university-led studies in mental health, musculoskeletal, and lifestyle medicine. ANZCTR is the authoritative source for academic research; ClinicalTrials.gov dominates for industry-sponsored oncology and cardiovascular trials. A complete picture of Australian clinical research requires both registries.
Sources: ANZCTR Statistics Tool (anzctr.org.au); Monash MUCTC Clinical Trial Trends 2023; MTPConnect Australia's Clinical Trials Sector Report 2024; Seidler et al. (2023) PMC10952960.
ASX Biotech Tracker
Live share prices and market data for ASX-listed Australian biotech companies with active clinical trial programs. Data sourced from Yahoo Finance via server-side proxy. Refreshes every 5 minutes.
ASX-Listed Companies with Australian Trial Activity
Neuren Pharmaceuticals
Paradigm Biopharmaceuticals
Live data via Yahoo Finance. Prices in AUD. 15–20 min delay may apply. Not financial advice.
Sector Overview
~100
ASX-listed life science companies
AU$100B+
Combined market capitalisation
#2
Global ranking for public biotech companies
Live Snapshot
Contact Us
Have a question about this dashboard, the data, or a potential collaboration? Get in touch with the SciEngage team.
SciEngage
Clinical Trials Intelligence
We typically respond within 1–2 business days. For urgent data inquiries, please include your organisation and the specific dataset or section you're referencing.
Privacy: This form uses your device's email client to send messages — no data is collected, stored, or processed by this website. Your name, email, and message are composed locally and only transmitted when you send the email from your own mail application. SciEngage does not use cookies, tracking pixels, or third-party analytics on this form. We will only use your contact details to respond to your inquiry and will not share them with any third party.